- +91 9403890582
- info@bcri.in
- Mon - Fri: 9:30 - 18:30
Welcome to the complete guide on pharmacovigilance. This tutorial provides detailed knowledge ideal for beginners, healthcare professionals, or anyone interested in drug safety careers.
Pharmacovigilance, also known as drug safety, is a critical discipline in healthcare focused on monitoring, evaluating, and preventing the harmful effects associated with medicines. Derived from the Greek word “pharmakon” (medicine or drug) and the Latin “vigilare” (to keep watch), pharmacovigilance literally means “to keep watch over drugs.”
This practice involves systematically collecting, analysing, and interpreting data about adverse drug reactions (ADRs), medication errors, and other potential drug-related problems. Its goal is to ensure that medicinal products remain safe and effective throughout their entire lifecycle—from the early stages of clinical research until they are available widely to the general public.
Every medication comes with potential risks, which may vary widely among different people. Some risks become apparent only after the drug has been used extensively in diverse populations. Pharmacovigilance plays a vital role in:
Pharmacovigilance isn’t just a national concern—it’s a global effort. Health regulatory authorities like the Food and Drug Administration (FDA) in the United States, the European Medicines Agency (EMA) in Europe, and the Pharmacovigilance Programme of India (PvPI) collaborate internationally to share vital drug safety data.
Organizations such as the World Health Organization (WHO) coordinate global databases like VigiBase, collecting and sharing information to ensure medicines worldwide meet rigorous safety standards.
Pharmacovigilance as a formal science is relatively new, but its importance has grown rapidly over the last few decades—especially due to real-world incidents that revealed the risks of using medicines without proper monitoring.
In the late 1950s and early 1960s, a drug called thalidomide was prescribed to pregnant women to treat morning sickness. However, it caused severe birth defects in thousands of babies across Europe and other parts of the world. This became one of the most tragic drug safety failures in medical history.
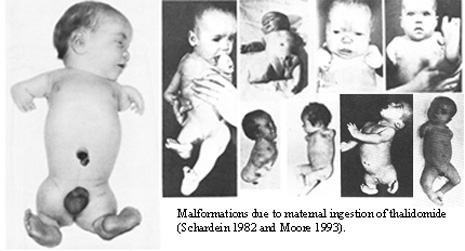
This disaster became a turning point and highlighted the urgent need for monitoring the safety of medicines, even after they were approved and released in the market.
After the thalidomide incident, governments and health organizations around the world began building systems to improve drug safety.
Some key developments include:
Today, more than 130 countries contribute data to VigiBase, the world’s largest global drug safety database maintained by WHO.
India launched its national pharmacovigilance system called PvPI (Pharmacovigilance Programme of India) in 2010, under the Central Drugs Standard Control Organization (CDSCO).
Key highlights of PvPI:
Thanks to PvPI, India has taken a strong role in ensuring medicines used across the country are monitored for safety—even after approval.
Pharmacovigilance is not just about collecting adverse event reports— it’s a structured process that helps detect, analyze, and prevent medication-related risks at every stage. Let’s break down the process into simple steps:
The first step in pharmacovigilance is to collect safety data. This includes reports of:
Data can come from:
In India, this is often reported using PvPI’s ADR reporting forms.
Once the data is collected, it needs to be entered into a drug safety database like:
At this stage, the report is:
This step ensures that the report is ready for medical review and can be used in signal analysis.
A trained medical reviewer or safety officer then examines the case to understand:
This step is called causality assessment, and it plays a big role in determining if the drug needs further investigation.
When multiple cases report similar events, analysts look for patterns or safety signals — unusual or unexpected side effects.
Tools like VigiBase (WHO’s global safety database) or internal analytics software help identify:
If a pattern is found, it’s flagged for investigation.
If a real risk is identified, the next step is to control or minimise that risk.
This may involve:
These actions are guided by Risk Management Plans (RMPs) developed by pharmaceutical companies and approved by regulators.
Finally, the findings are communicated to:
Companies may need to submit regular reports like:
This step helps keep the healthcare system informed and ensures everyone uses medicines safely.
To understand pharmacovigilance clearly, it’s important to get familiar with some commonly used terms. Here’s a simplified glossary with practical examples to help you learn the language of drug safety:
🔹 Definition: A harmful or unintended response to a medicine at normal doses.
Example: A patient takes an antibiotic for a throat infection and develops a severe skin rash. This unexpected reaction is an ADR.
ADRs are the core focus of pharmacovigilance. They can range from mild (e.g., nausea) to serious (e.g., liver failure). 2.
🔹 Definition: Any unfavorable medical occurrence that happens after taking a drug, whether or not it’s caused by the drug.
🔹 Example: A person gets a headache after taking a vitamin supplement. It may or may not be related to the supplement—this this is an AE.
All ADRs are AEs, but not all AEs are ADRs. The link with the drug must be evaluated.
🔹 Definition: An adverse event that results in death, is life-threatening, causes hospitalization or disability, or leads to birth defects.
🔹 Example: A patient develops severe breathing problems and is hospitalized after taking a new asthma inhaler. This is classified as an SAE.
🔹 Definition: Information that suggests a new potential risk from a drug, requiring further evaluation.
🔹 Example: If many patients report high blood pressure after using a new diabetes medicine, this might trigger a safety signal.
Signals do not confirm danger-they indicate the need for deeper investigation.
🔹 Definition: The process of determining whether a drug caused an adverse reaction.
🔹 Example: If a patient takes a painkiller and develops ulcers, doctors will evaluate the timing, dosage, and other factors to decide if it’s likely due to the drug.
Tools like WHO-UMC criteria or the Naranjo Algorithm are commonly used for assessment.
🔹 Definition: A global medical terminology used to standardize how drug safety terms are recorded and analyzed.
🔹 Example: Instead of writing “stomach pain,” the system uses a MedDRA term like “Abdominal pain upper” to keep data consistent.
MedDRA ensures that drug safety data from around the world can be compared and analyzed efficiently.
🔹 Definition: Voluntary reporting of adverse events by healthcare professionals or patients.
🔹 Example: A doctor notices a skin reaction in a patient using a new cream and sends a report to the PvPI system. This is a spontaneous report.
This is a key source of data for early detection of drug-related risks.
🔹 Definition: A document created by pharmaceutical companies to outline how they will monitor and manage risks related to their products.
🔹 Example: A new cancer drug may come with an RMP requiring regular blood tests to monitor liver function.
RMPs are submitted to regulatory authorities before and after product launch.
🔹 Definition: A document that describes a company’s entire pharmacovigilance system, including roles, responsibilities, procedures, and databases.
🔹 Example: If a pharma company is audited, the PSMF shows how they track and report safety data.
🔹 Definition: Regular reports submitted to regulators containing updated safety data and benefit–risk analysis of a drug.
🔹 Example: Every 6 months or 1 year, companies submit a PSUR/PBRER for drugs under surveillance to show updated safety information and any action taken.
🔹 Definition: A legally responsible person appointed by a pharmaceutical company to ensure compliance with all drug safety requirements.
🔹 Example: The QPPV ensures that all ADRs for a particular drug are reported to the regulatory authorities within the required timelines.
In India, this role may be handled by drug safety officers or Regulatory Affairs professionals.
🔹 Definition: The global ADR database managed by the WHO, which collects reports from over 130 countries.
🔹 Example: PvPI in India sends adverse event data to VigiBase, helping monitor drug safety worldwide.
Term | Full Form |
ADR | Adverse Drug Reaction |
AE | Adverse Event |
SAE | Serious Adverse Event |
RMP | Risk Management Plan |
PBRER | Periodic Benefit-Risk Evaluation Report |
PSMF | Pharmacovigilance System Master File |
QPPV | Qualified Person for Pharmacovigilance |
The drug safety lifecycle explains how pharmacovigilance works at every stage of a medicine’s journey— from testing in the lab to being used by real-world patients. Drug safety doesn’t stop after approval-in in fact, it becomes even more important once the drug is on the market.
Let’s understand how drug safety is monitored during two major stages: pre-approval and post-approval.
Before a new medicine can be sold to the public, it goes through a series of controlled studies called clinical trials.
These are divided into three phases:
💡 Note: If a drug fails at any of these phases due to safety concerns, it may never reach the market.
Once a drug is approved and released to the market, it enters Phase IV, also known as post-marketing surveillance.
📌 Key Activities Include:
🚨 Some drugs have been recalled or restricted during Phase IV due to rare but serious side effects only seen after widespread use.
🧠 In Summary
Stage | What Happens | Who’s Involved |
Pre-Approval | Safety & efficacy tested in clinical trials | Pharma companies, CROs, regulators |
Post-Approval | Real-world safety monitoring | Patients, doctors, regulatory authorities |
The drug safety lifecycle is continuous and dynamic. Pharmacovigilance plays a vital role at every phase to ensure that medicines stay safe and effective throughout their time on the market.
Enroll in a clinical research & pharmacovigilance training program today and take the first step toward a rewarding career!
Pharmacovigilance is not a single task—it’s a series of well-defined activities that together ensure drug safety across the lifecycle of a medicine. Below are the seven core activities carried out by pharmacovigilance professionals, regulatory agencies, and pharma companies:
✅ What it means:
When a new adverse event (AE) or adverse drug reaction (ADR) is reported—by a doctor, patient, or pharmacist – the first step is to receive (intake) the case and sort (triage) it.
⚙️ What happens:
🧾 Example:
A hospital reports a patient death possibly linked to a new painkiller. This case is triaged as serious and expedited, requiring fast follow-up.
✅ What it means:
Once a case is accepted, all details are entered into a pharmacovigilance database, and medical terms are coded using MedDRA (Medical Dictionary for Regulatory Activities).
⚙️ What happens:
🧾 Example:
If a patient reports “upset stomach,” it is coded as “Dyspepsia” under MedDRA. This ensures every report uses uniform medical language.
✅ What it means:
Trained medical reviewers evaluate whether the reported reaction was likely caused by the drug — a process known as causality assessment.
⚙️ What happens:
🧾 Example:
A patient develops liver damage after using a medication. Medical experts assess if the liver issue is likely related to the drug or due to another disease.
✅ What it means:
Companies must compile summary reports of safety data collected over time— not just individual cases.
⚙️ What happens:
🧾 Example:
A PSUR may report that no new risks were identified in the last 6 months of drug use — helping regulators decide if label changes are needed.
✅ What it means:
A signal is a trend or pattern in the data that suggests a new safety concern. Pharmacovigilance teams detect these by analyzing reports in bulk.
⚙️ What happens:
🧾 Example:
If multiple people report seizures after taking a new cough syrup, this pattern could trigger a safety signal that needs urgent attention.
✅ What it means:
A Risk Management Plan (RMP) is a detailed document that outlines how a company will monitor and minimize known or potential risks of its product.
⚙️ What happens:
🧾 Example:
A cancer drug that may cause heart issues might include ECG monitoring in its RMP.
✅ What it means:
Once a risk is identified or confirmed, it must be communicated clearly to doctors, pharmacists, and the public — and reported to regulators within set timelines.
⚙️ What happens:
🧾 Example:
If a new side effect is added to a drug’s label, all doctors and pharmacists are informed via safety letters or electronic updates.
✅ In Summary
Step | Activity | Key Purpose |
1 | Case Intake & Triage | Receive and sort incoming reports |
2 | Data Entry & Coding | Enter cases using standard MedDRA terms |
3 | Medical Review | Assess seriousness and drug-event link |
4 | Aggregate Reports | Submit regular safety summaries (PSUR, DSUR) |
5 | Signal Detection | Spot patterns indicating new risks |
6 | Risk Management | Develop plans to monitor and minimize risks |
7 | Risk Communication | Alert stakeholders and regulatory agencies |
Pharmacovigilance isn’t just a best practice—it’s a legal requirement. Every country has its own rules and regulations to ensure that medicines are monitored for safety throughout their life cycle. Pharmaceutical companies must follow these regulations to protect public health and stay compliant with the law.
Here’s a beginner-friendly overview of major global and Indian pharmacovigilance guidelines:
The ICH brings together regulatory authorities and pharmaceutical companies to develop common standards for drug safety.
💡 These guidelines ensure consistency in how drug safety is managed across countries like the USA, EU, Japan, and more.
💡 In the EU, companies must appoint a Qualified Person for Pharmacovigilance (QPPV) who is legally responsible for drug safety.
📋 Key Features of PvPI:
📝 Indian Regulatory Requirements for Companies:
💡 CDSCO may issue notices, withdraw licenses, or take legal action for non-compliance.
Following pharmacovigilance regulations helps to:
✅ In Summary
Region | Guiding Body | Key Guidelines |
Global | WHO / ICH | ICH E2E, E2D, VigiBase |
USA | FDA | MedWatch, 15-day reporting, REMS |
Europe | EMA | GVP Modules, QPPV role |
India | CDSCO / PvPI | ADR Reporting, PSURs, PSMF |
Pharmacovigilance is a team effort. Different professionals work together to ensure the safety of medicines by collecting, reviewing, analyzing, and reporting adverse drug reactions (ADRs). Below are the most common job roles in a pharmacovigilance system, along with their core responsibilities.
The QPPV is the most senior and legally responsible person for all drug safety matters in a pharmaceutical company, especially in the European Union (EU) and other regulated markets.
🛠️ Key Responsibilities:
📌 In India, the QPPV role is usually handled by a Medical Officer or Regulatory Affairs Head, depending on company size.
✅ Who is a DSA?
A Drug Safety Associate is usually the first point of contact in the case processing team. It is a popular entry-level role in pharmacovigilance for life science graduates.
🛠️ Key Responsibilities:
💼 Ideal for freshers in pharmacy, microbiology, biotech, or life sciences.
✅ Who is a PV Officer?
A Pharmacovigilance Officer supervises the safety team’s daily operations and ensures smooth workflow in case processing, compliance, and communication.
🛠️ Key Responsibilities:
📌 This is a mid-level role often held by professionals with 2–4 years of experience.
✅ Who is a Safety Data Analyst?
This role focuses on interpreting pharmacovigilance data using tools and analytics to identify trends, patterns, and safety signals.
🛠️ Key Responsibilities:
📈 A good fit for candidates with data analytics or biostatistics background.
✅ Who is a Medical Reviewer?
A Medical Reviewer (usually an MBBS, MD, BDS, or PharmD graduate) evaluates the clinical significance of adverse events and provides medical judgement.
🛠️ Key Responsibilities:
🧠 This role requires strong medical knowledge and clinical judgement.
🧠 In Summary:
Role | Key Focus | Ideal Background |
QPPV | Legal responsibility, compliance | Senior with regulatory/PV expertise |
Drug Safety Associate | Case intake, data entry, MedDRA coding | Life sciences, pharma freshers |
Pharmacovigilance Officer | Supervision, SOPs, audit readiness | 2–4 years PV experience |
Safety Data Analyst | Data analysis, signal detection | Analytics, statistics, PV |
Medical Reviewer | Clinical evaluation of cases | MBBS, MD, BDS, PharmD |
Pharmacovigilance is a global effort. Since medicines are often used across many countries, drug safety monitoring must be a shared responsibility. Countries, regulatory authorities, and international health organizations work together to track adverse effects, issue warnings, and protect patients worldwide.
The World Health Organization (WHO) launched the Programme for International Drug Monitoring (PIDM) in 1968 following the thalidomide tragedy.
Today, more than 130 countries are part of this program, contributing safety data and working together to:
🧠 India is an active member of PIDM through PvPI, and contributes reports regularly to WHO.
VigiBase is the world’s largest database of adverse drug reactions, managed by the Uppsala Monitoring Centre (UMC) in Sweden on behalf of WHO.
✅ Key Facts:
🌐 Example: If several countries report a rare side effect for a new medicine, VigiBase helps spot the trend early and alerts the global community.
The EMA oversees medicine safety in the European Union. It introduced the Good Pharmacovigilance Practices (GVP) — a detailed set of modules that guide every part of drug safety.
🔍 GVP Modules Include:
Each EU-based pharma company must appoint a Qualified Person for Pharmacovigilance (QPPV) who is legally responsible for ensuring compliance.
The Food and Drug Administration (FDA) monitors drug safety in the United States through:
💡 The FDA mandates 15-day reporting for serious and unexpected adverse events.
Japan monitors pharmacovigilance through the PMDA, which collects adverse event reports, especially for:
Japan is also an active ICH member and aligns with global standards.
📘 ICH (International Council for Harmonisation)
The ICH develops unified technical guidelines used in the USA, Europe, Japan, and many other countries. Key pharmacovigilance guidelines include:
🧪 CIOMS (Council for International Organizations of Medical Sciences)
CIOMS works with WHO to develop best practices in global drug safety. Its publications guide ethical standards and reporting methods in PV.
📌 Summary Table
Region | Organization | Key Tools/Programs |
🌐 Global | WHO, UMC | VigiBase, PIDM |
🇪🇺 Europe | EMA | GVP, EudraVigilance, QPPV |
🇺🇸 USA | FDA | MedWatch, FAERS, REMS |
🇮🇳 India | CDSCO / PvPI | VigiFlow, Mobile ADR App |
🇯🇵 Japan | PMDA | PMDA Reporting System |
🌍 International | ICH, CIOMS | E2 series guidelines, ethical PV |
Pharmacovigilance involves managing large volumes of safety data, and this is only possible through the use of specialized tools and databases. These tools help collect, process, analyze, and report adverse events in a structured and compliant manner.
Let’s explore the most widely used pharmacovigilance tools and databases, both globally and within companies.
🌐 India contributes to VigiBase through VigiFlow under PvPI.
In India, ADR Monitoring Centres (AMCs) enter data into VigiFlow, which is reviewed by the Indian Pharmacopoeia Commission (IPC).
🔐 Argus ensures compliance with FDA, EMA, and ICH reporting standards.
Preferred by mid-sized pharma companies and outsourcing agencies.
Used for signal detection and monitoring benefit–risk balance in the EU.
Example: “Dizziness” is coded uniformly as a Preferred Term (PT) in MedDRA across all safety reports.
Other commonly used PV tools include:
As pharmacovigilance becomes more data-driven, companies also use:
These tools help in signal detection, trend analysis, and improving decision-making speed.
✅ In Summary:
Tool / Database | Used For | Organization |
VigiBase | Global ADR data collection | WHO / UMC |
VigiFlow | National-level ADR submission | PvPI / WHO |
Argus Safety | End-to-end PV automation | Oracle |
ARISg | Cloud-based case processing | ArisGlobal |
EudraVigilance | EU ADR database | EMA |
MedDRA | Coding of medical terms | MSSO |
Power BI / SAS / AI | Analytics and visualization | Industry-wide |
Pharmacovigilance has grown into a critical part of modern healthcare. But as new drugs, technologies, and patient expectations evolve, the field faces both challenges and opportunities. Let’s explore the current issues and future trends shaping the world of drug safety.
Problem: Most ADRs go unreported — especially in developing countries.
📌 Impact: Delays in detecting serious safety issues.
Problem: Pharmacovigilance systems receive millions of reports annually.
📌 Impact: Important safety signals may be missed in a sea of low-quality data.
Problem: Different countries have different rules, reporting systems, and formats.
📌 Impact: Difficulties in combining and comparing global safety data.
Problem: Biologics, gene therapies, and combination drugs require new safety frameworks.
📌 Impact: Additional training and tools are needed to monitor new drug classes.
Problem: Today, patients discuss side effects on social media, apps, and online forums.
📌 Impact: Missed signals in informal but valuable platforms.
💡 Example: Machine learning models can identify high-risk cases from thousands of reports in minutes.
💡 Example: Tracking long-term safety of diabetes drugs using hospital records.
💡 Example: Some cancer drugs are prescribed only after genetic screening to minimize risks.
💡 Patients and caregivers can now report side effects without needing to visit a hospital.
💡 Example: Some countries now require Risk Management Plans (RMPs) even before drug approval.
🧠 Summary: Challenges vs Trends
Challenges | Emerging Solutions |
Underreporting | Mobile apps, awareness campaigns |
Data overload | AI-powered automation |
Lack of standardization | Global harmonization (ICH, WHO) |
New complex drugs | Advanced PV tools, specialized training |
Social media monitoring | AI, real-time listening tools |
As the global pharmaceutical industry grows, so does the need for professionals who can ensure the safety of medicines. Pharmacovigilance (PV) has become a promising and in-demand career option for graduates in life sciences, pharmacy, biotechnology, and medicine.
Drug Safety Associate (DSA)
Pharmacovigilance Executive / Officer
Case Processor / ICSR Associate
📈 Mid-Level & Senior Roles
Medical Reviewer
Safety Data Analyst
Aggregate Report Specialist
Signal Manager / Risk Management Expert
🎓 Skills Needed to Succeed in Pharmacovigilance
India | International |
TCS, Cognizant, Accenture, IQVIA | Pfizer, Roche, GSK, Bayer |
Parexel, Bioclinica, Novotech | Parexel, PRA Health Sciences |
Lambda, CliniRx, APCER | ICON, Covance, Syneos Health |
Enroll in a clinical research & pharmacovigilance training program today and take the first step toward a rewarding career!
Pharmacovigilance is more than just monitoring side effects — it’s about protecting lives, building trust in medicine, and supporting innovation in healthcare. From clinical trials to post-marketing surveillance, pharmacovigilance ensures that every drug used by patients remains as safe and effective as possible.
As the world continues to adopt new therapies and technologies, the demand for skilled pharmacovigilance professionals is rising rapidly. Whether you’re a student, life science graduate, or healthcare worker, this field offers a rewarding, future-proof career path filled with purpose and global opportunities.
At BCRI, we’re committed to helping you enter and succeed in this exciting field. With industry-aligned training, practical case study exposure, and career support — you’re one step closer to becoming a trusted guardian of drug safety.
Join thousands of life science graduates who have successfully launched their careers in drug safety with the right training.
👉 Enroll in BCRI’s Pharmacovigilance Certification Program
🎓 Your career in clinical drug safety starts here.
WhatsApp us


Recognized Training Partner under Life Sciences Sector Skill Development Council
Live mentor support • Internship & placement assistance*
Our Team would love to Guide you to select the right course here .
